
Rischin: Travel/Accommodation/Expenses: Merck Research grant/Funding (self): Genentech/Roche, Merck, Regeneron, Bristol-Myers Squibb, GlaxoSmithKline, Sanofi. Greil: Advisory/Consultancy: Celgene, Novartis, Roche, Bristol-Myers Squibb, Takeda, AbbVie, AstraZeneca, Janssen, MSD, Merck, Gilead Sciences, Daiichi Sankyo Travel/Accommodation/Expenses: Rocher, Amgen, Janssen-Cilag, AstraZeneca, Novartis, MSD, Celgene, Gilead Sciences, Bristol-Myers Squibb Honoraria (self): Celgene, Roche, Merck, Takeda, AstraZeneca, Novartis, Amgen, Bristol-Myers Squibb, MSD, Sandoz, AbbVie, Gilead Sciences, Daiichi Sankyo Research grant/Funding (institution): Celgene, Merck, Takeda, AstraZeneca, Novartis, Amgen, Bristol-Myers Squibb, MSD, Sandoz, Gilead Sciences, Roche. Merck Sharp & Dohme Corp., a subsidiary of Merck & Co., Inc., Kenilworth, NJ, USA. 
This assistance was funded by Merck Sharp & Dohme Corp., a subsidiary of Merck & Co., Inc., Kenilworth, NJ, USA. Medical writing and editorial assistance was provided by Doyel Mitra, PhD, CMPP, and Jo Bairzin, PhD, of the ApotheCom pembrolizumab team (Yardley, PA, USA). *Nominal values not adjusted for multiplicity. 
Safety was favorable for pembro vs E and comparable for pembro+C vs E Table: 915MO Long-term follow-up confirmed the statistically significant improvement in OS established at the protocol-specified interim and final analyses for pembro vs E in pts with PD-L1 CPS ≥20 and CPS ≥1 and for pembro+C vs E in pts with PD-L1 CPS ≥20 and CPS ≥1, and total pop. In all, 11 pts (pembro, 6 pembro+C, 5) received a secondcourse of pembro ORR for second course was 36.4% (CR, 1 PR, 3). Treatment-related grade 3-5 AEs: 17.0% for pembro, 71.7% for pembro+C, and 69.3% for E. OS, PFS, ORR, and DOR data are shown in the table. Pembro and pembro+C improved OS vs E in the CPS ≥20, CPS ≥1, and total pops (Table). Median study follow-up (March 25, 2020) was 46.2 mo for pembro vs E and 45.6 mo for pembro+C vs E. Efficacy was analyzed in the ITT pop safety was analyzed in all randomized pts who received ≥1 dose of study drug. 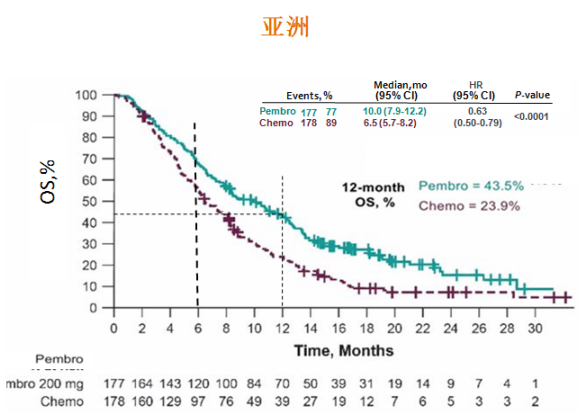
Follow-up was defined as time from randomization to database cutoff. Pts in the pembro or pembro+C arms who stopped pembro with stable disease or better and then had PD could receive a second course of pembro if eligible. Pts with locally incurable R/M HNSCC were randomized to 1L pembro 200 mg Q3W for 24 mo, pembro+C, or E. Safety was favorable for pembro vs E and comparable for pembro+C vs E. Pembro+C significantly prolonged OS vs E in the PD-L1 CPS ≥20, CPS ≥1, and total pops. In the phase III KEYNOTE-048 study, pembro significantly prolonged OS vs E in patients (pts) with PD-L1 combined positive score (CPS) ≥20 and CPS ≥1 and had noninferior OS in the total population (pop).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
